Immunodermatology
AG Schmieder
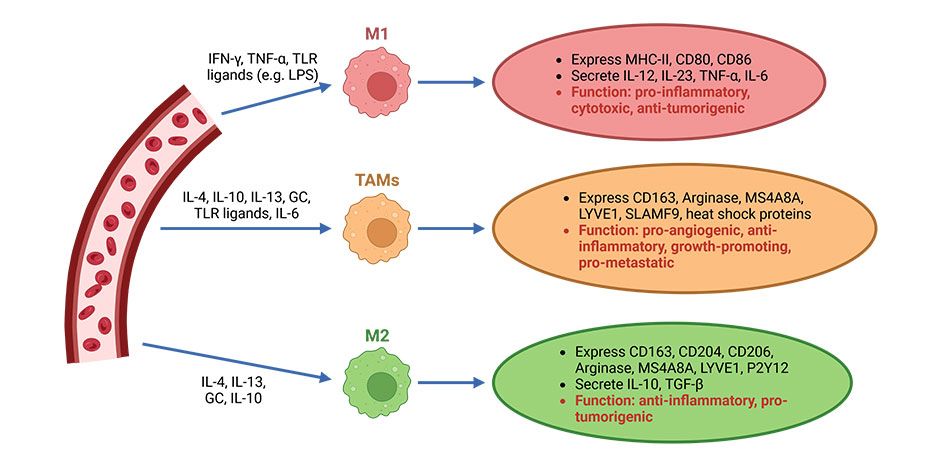
Macrophages are central effector cells of the innate immune system and play a role in many pathological processes. They are divided into pro-inflammatory (M1) and anti-inflammatory (M2) macrophages. These two macrophage phenotypes differ in the expression of their cell surface molecules, secreted cytokines, and biological functions. It has been shown that the respective M1 and M2 subtypes can be reversibly manipulated, leading to altered macrophage functionality. Exactly which signaling pathways are used to achieve these changes and what impact this has on skin diseases remains to be elucidated.
Skin cancer is one of the five most common cancers worldwide. Once skin cancer metastasizes, the 5-year survival rate drops down to 60%. The introduction of immune checkpoint inhibitors (ICI) significantly improved the prognosis of stage IV melanoma patients, however, about 40-50% of patients do not respond to ICI. In addition, every second patient develops immune-related side effects or an intrinsic resistance to therapy.
Tumor-associated macrophages (TAM) are partly responsible for intrinsic resistance to ICI, as these cells promote immunosuppression, melanoma growth and metastasis. It has been shown that resistance to PD-1 immunotherapy is associated with increased infiltration of M2-like TAM. In this context, we are investigating the possibility of repolarization of M2-like TAM to a proinflammatory M1-like phenotype. We hope that this will provide new insights into the mechanisms of treatment resistance while identifying novel therapeutic approaches.
In our group, both in vivo models and primary in vitro cultures are used to analyze the underlying mechanisms. Common methods such as qRT-PCR, FACS, Western blot, ELISA and immunochemical staining are complemented by modern analytical techniques such as AI-based quantification of data or single cell sequencing.
We are always happy to receive unsolicited applications from motivated students and technical assistants.
If you are interested to work with us, please send your comprehensive application documents to: schmieder_a@ukw.de
Sources
Yunna C, Mengru H, Lei W & Weidong C.
Macrophage M1/M2 polarization.
Eur J Pharmacol: 2020; 877:173090.
Schmieder A, Michel J, Schonhaar K, Goerdt S & Schledzewski K.
Differentiation and gene expression profile of tumor-associated macrophages.
Semin Cancer Biol: 2012; 22:289-97.
Aras S, Zaidi MR.
TAMeless traitors: macrophages in cancer progression and metastasis.
Br J Cancer: 2017; 117:1583-91.
Larkin J, Chiarion-Sileni V, Gonzalez R, Grob JJ, Rutkowski P, Lao CD, Cowey CL, Schadendorf D, Wagstaff J, Dummer R, Ferrucci PF, Smylie M, Hogg D, Hill A, Marquez-Rodas I, Haanen J, Guidoboni M, Maio M, Schoffski P, Carlino MS, Lebbe C, McArthur G, Ascierto PA, Daniels GA, Long GV, Bastholt L, Rizzo JI, Balogh A, Moshyk A, Hodi FS & Wolchok JD.
Five-Year Survival with Combined Nivolumab and Ipilimumab in Advanced Melanoma.
N Engl J Med: 2019; 381:1535-46.
Falleni M, Savi F, Tosi D, Agape E, Cerri A, Moneghini L& Bulfamante GP.
M1 and M2 macrophages' clinicopathological significance in cutaneous melanoma.
Melanoma Res: 2017; 27:200-10.
Selected publications
Kloss, L., Dollt, C., Schledzewski, K., Krewer, A., Melchers, S., Manta, C., Sticht, C., de la Torre, C., Utikal, J., Umansky, V, Schmieder, A. (2019).
ADP secreted by dying melanoma cells mediates chemotaxis and chemokine secretion of macrophages via the purinergic receptor P2Y12.
Cell Death Dis. 2019 Oct 7;10(10):760.
Michel, J., Dollt, C., Kloss, L. Melchers, S., Schledzewski, K., Becker, K., Sauer, A., Krewer, A., Koll, F., and Schmieder, A. (2018).
The novel immunoglobulin super family receptor SLAMF9 identified in TAM of murine and human melanoma influences pro-inflammatory cytokine secretion and migration.
Cell Death & Disease. CDDIS-18-0271R, accepted for publication.
Dollt, C., Becker, K., Michel, J., Melchers, S., Weis, C. A., Schledzewski, K., Krewer, A., Kloss, L., Gebhardt, C., Utikal, J., and Schmieder, A. (2017).
The shedded ectodomain of Lyve-1 expressed on M2-like tumor-associated macrophages inhibits melanoma cell proliferation.
Oncotarget, 103682-103692.
Michel, J., Schonhaar, K., Schledzewski, K., Gkaniatsou, C., Sticht, C., Kellert, B., Lasitschka, F., Geraud, C., Goerdt, S., and Schmieder, A. (2013).
dentification of the novel differentiation marker MS4A8B and its murine homolog MS4A8A in colonic epithelial cells lost during neoplastic transformation in human colon.
Cell death & disease, e469.
Schmieder, A., Schledzewski, K., Michel, J., Schonhaar, K., Morias, Y., Bosschaerts, T., Van den Bossche, J., Dorny, P., Sauer, A., Sticht, C., et al. (2012c).
The CD20 homolog Ms4a8a integrates pro- and anti-inflammatory signals in novel M2-like macrophages and is expressed in parasite infection.
European journal of immunology, 2971-2982.
Schmieder, A., Schledzewski, K., Michel, J., Tuckermann, J. P., Tome, L., Sticht, C., Gkaniatsou, C., Nicolay, J. P., Demory, A., Faulhaber, J., et al. (2011b).
Synergistic activation by p38MAPK and glucocorticoid signaling mediates induction of M2-like tumor-associated macrophages expressing the novel CD20 homolog MS4A8A.
International journal of cancer, 122-132
Contact
Appointment Policlinic
+49 931 201-26714
Monday to Thursday
12:00 PM to 16:00 PM
Friday
12:00 PM to 15:00 PM
Appointment In-patient care
Monday to Friday
08:00 AM to 12:00 PM
Margit Körner
+49 931 201-26354
Telephone
Gate
+49 931 201-26710
Directorate
Prof. Dr. med. Matthias Goebeler
Secretary's office
Gudrun Braun
+49 931 201-26351
Student matters
Ute Düchs
+49 931 201-26353
Address
Klinik und Poliklinik für Dermatologie, Venerologie und Allergologie | Josef-Schneider-Straße 2 | Haus D8 | 97080 Würzburg | Deutschland