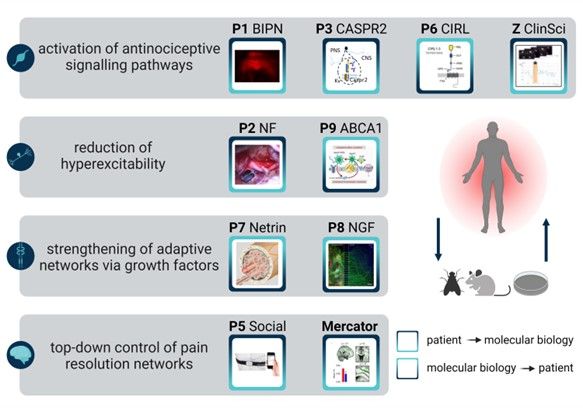
Top-down method: P1 to P5
Project leaders of the KFO are not only from different institutions, but also from both basic research and clinical settings. This interdisciplinary cooperation provides best conditions for efficient translational research. This will be approached from two directions: Projects P1 to P5 are primarily implemented in a top-down process, in which the patients are the starting point for the research question. Sample material is collected and analysed. Subsequently, mechanisms are tested on the animal model.
Bottom-up principle: P6 to P9
Projects P6 to P9, on the other hand, follow the bottom-up principle. Initial findings are acquired in cell models and model organisms such as flies and rodents in order to be transferred to patients in the following. For a successful exchange of information between clinic and research, project leaders as well as PainScouts and post docs meet at joint Jour Fixes, as well as in PainScouts training and the hands-on workshops.
Aims, mechanisms and methods
Within the framework of KFO 5001, various mechanisms of pain resolution are being investigated: Four projects are study increased activation of antinociceptive signalling pathways for pain resolution. Two projects focus on the reduction of hyperexcitability. Two other projects investigate basic mechanisms to support pain resolution by growth factors. Special imaging techniques and smartphone techniques are used to monitor the pain in daily activities.
Enhancement of antinociceptive signalling pathways
P1 – Bortezomib-induced polyneuropathy
In this project, we postulate that pain resolution in patients with bortezomib-induced polyneuropathy is governed by the release of anti-inflammatory cytokines. We acquire an in-depth clinical examination of the patients and study the genetic predisposition as well as changes in the blood and in the skin. These could be biomarkers for the course of the disease.
P3 – Anti-Caspr2 autoantibodies
The P3 project deals with autoantibodies against nerve structures that initially cause nerve pain but then regress via immune mechanisms during the healing process. We want to decipher their exact mode of action and their influence, especially on certain ion channels of the spinal ganglia.
P6 – Adhesion GPCR CIRL
In project P6, the function of CIRL as a so-called antinociceptive G-protein coupled receptor on the cell membrane of nerve cells is being investigated. CIRL can be activated by mechanical stimuli. Therefore, we want to explore the detailed mechanisms and processes of the pain-relieving role of CIRL in more detail and find out how best to activate CIRL for pain resolution.
PZ – Cytokine signature and lipid homeostasis
The service project Z involves the identification of the cytokine signature in the blood and the analysis of lipid homeostasis. From the centrally collected biosamples and data, new examination methods are to be established and, in particular, relevant and objective markers for the course of pain are to be determined.
Reduction of hyperexcitability
P2 – Gene-based neuropathies
Project P2 focuses on pathological nerve alterations in neurofibromatosis (NF) as an example of gene-based neuropathies. The underlying gene mutations lead to hyperexcitability of pain receptors in animal models.
P9 – ABC transporter ABCA1
The influence of cholesterol, oxidised phospholipids, and the corresponding transport proteins on pain resolution are examined in project P9. It is known that oxidised phospholipids activate specific ion channels and thus lead to the excitation of pain receptors. Apolipoprotein components, i.e., the transport proteins of the fat molecules, can prevent this process.
Strengthening networks with growth factors
P7 – Netrin and nerve barriers
Project 7 focuses on the sealing of the blood-nerve barrier by the endogenous growth factor netrin-1 as a prerequisite for pain resolution. We are investigating the regenerative effect of netrin on leaky barriers protecting nerves, as well as the therapeutic applicability of netrin in neuropathic pain.
P8 – Neurotrophic factors
Project P8 investigates the influence of nerve growth factors on protein synthesis in the axon during the resolution phase of pain. This in turn influences the excitability of the neurons. It will be investigated which messenger substances and which changes in the messenger RNA (mRNA) are decisive in this process.
Control of pain resolution networks
P5 – Influence of the social environment on pain
Project P5 focuses on the influence of the social environment and corresponding changes in the brain on pain perception and its resolution. In addition, pain resolution networks are evaluated using top-down control. Using smartphone surveys, we are trying to record the social life of those affected and compare it with the subjective perception of pain.
Mercator Fellow – Imaging techniques
In collaboration with Prof. Paul Geha MD from the University of Rochester, changes in the brain of patients with chronic pain conditions are being investigated using imaging techniques. In long-term monitoring, this can be assigned to the group of pain regression or pain persistence, and biomarkers or predictors can also be found.


