
June 2025
Automated segmentation of the dorsal root ganglia in MRI.

Nauroth-Kreß AC, Weiner S, Hölzli L, Kampf T, Homola GA, Pham M, Kollmannsberger P, Schindehütte M.
Neuroimage. 2025 May 1;311:121189. doi: 10.1016/j.neuroimage.2025.121189. Epub 2025 Apr 2. PMID: 40185423.
Rationale
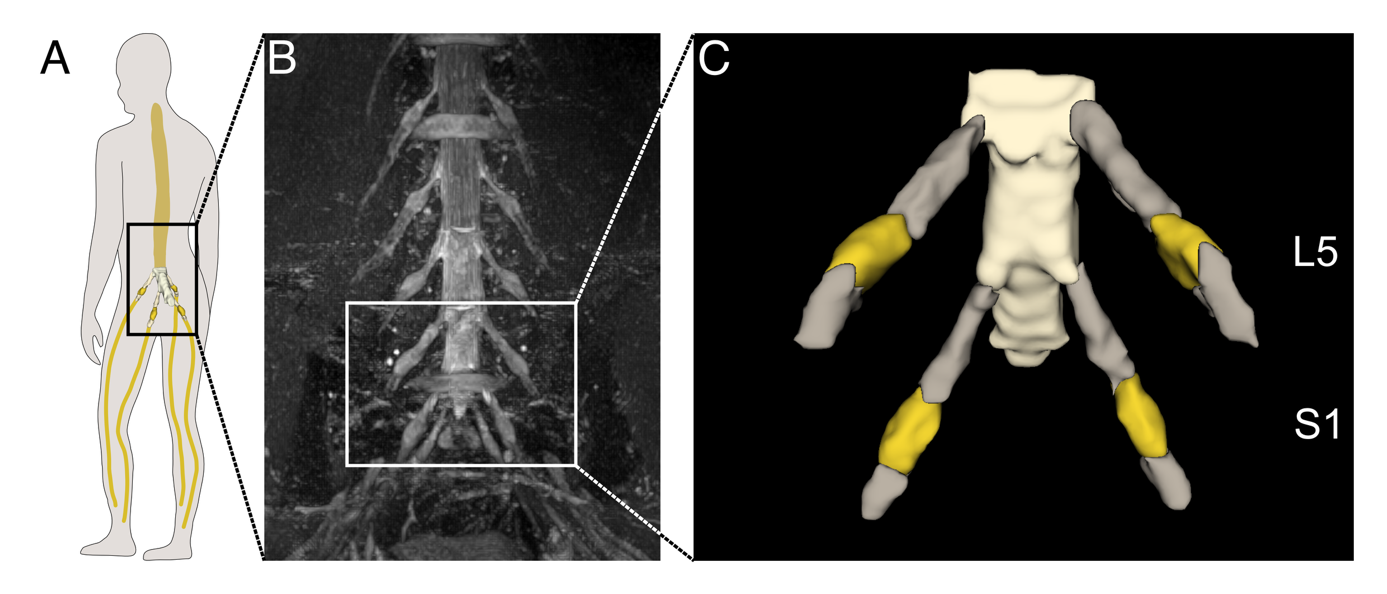
The dorsal root ganglia (DRG) are small structures of the peripheral nervous system that house sensory neurons and are thought to play a key role in the development and processing of pain. Recent advances in Magnetic Resonance Imaging (MRI) now make it possible to visualize the DRG in living individuals, with features such as volume and signal intensity emerging as promising imaging biomarkers for conditions like neuropathic pain. However, the DRG’s small size poses significant challenges for non-invasive imaging. At present, analysis of DRG MRI relies on manual segmentation by experts - an approach that is time-consuming and prone to variability. To enable the broader use of DRG imaging in larger studies and across clinical sites, there is an urgent need for fast, reliable, and fully automated methods for image analysis.
Experimental Design and Results
To overcome the limitations of manual DRG analysis, the researchers developed a fully automated segmentation method using deep learning. They first created reference data by having three experts manually segment lumbosacral DRGs in MRI scans of healthy individuals. These annotations were combined into a single “ground truth” to reduce bias. A deep learning model (nnU-Net) was then trained on these annotated scans and tested on independent MRI data from both healthy subjects and patients with Fabry disease, a condition known to affect the DRG. Finally, the team compared DRG volume and signal intensity measurements derived from the automated segmentations to those from expert annotations. The automated model achieved accuracy comparable to human experts while reducing processing time from 15–20 minutes to just 100 seconds per image. It also successfully detected clinically relevant differences in DRG signal intensity between healthy males and Fabry patients with pain, demonstrating both precision and potential clinical utility.
Significance and Perspective
This study presents the first fully automated method for 3D segmentation of the dorsal root ganglia (DRG) in MRI using deep learning. The approach achieves expert-level accuracy while being substantially faster and more consistent. This advance is a critical step toward enabling large-scale, objective analysis of DRG structure and function, both within single centers and across multiple clinical sites. By providing a reliable and reproducible way to assess DRG changes, the method opens new possibilities for using DRG imaging as a biomarker in conditions such as neuropathic pain. It holds promise for improving our understanding of disease mechanisms, tracking progression over time in individual patients, and evaluating treatment effects. Although further validation in a broader range of pathologies is needed, this tool marks a significant advancement for both clinical research and the study of the peripheral nervous system in pain disorders.
Further Information
Dr. Magnus Schindehütte, MD, is an Advanced Clinician Scientist and Principal Investigator of the Clinical Research Unit KFO5001, ResolvePAIN. Together with Prof. Rittner and Prof. Krug, he leads project NP7.
Previous Paper of the Month
April 2025
Autoantibodies in patients with fibromyalgia syndrome.
Seefried S, Barcic A, Grijalva Yepez MF, Reinhardt L, Appeltshauser L, Doppler K, Üçeyler N, Sommer C.
Pain. 2025 Feb 5. doi: 10.1097/j.pain.0000000000003535. Epub ahead of print. PMID: 39907533.
Background
Fibromyalgia syndrome (FMS) is a chronic condition characterized by widespread pain, sleep disturbances, fatigue, and often psychological symptoms. The exact causes are still unclear. While was once considered a rheumatological disease, later disturbed central pain processing was assumed to be the main mechanism by most researchers. The present study provides evidence for an additional peripheral pathomechanism and identifies the involvement of the immune system in a subgroup of patients.
Experimental Design and Results
In this study, serum from patients with fibromyalgia syndrome was tested for autoantibodies - immune system defense proteins that target the body's own structures. Using dorsal root ganglion (DRG) sections for binding assays, autoantibodies were detected in over 35% of the samples.
Different patterns were identified among individual patients, such as predominant binding to large DRG nerve cells responsible for transmitting sensory information, or binding to satellite cells, which surround and support these nerve cells. The type of binding was associated with patients’ symptoms.
Significance and Perspective
The results suggest that, in some affected individuals, autoantibodies are present and might contribute causally to the pathophysiology of FMS. Future research will aim to clarify which specific target structures are attacked. This could lead to new therapeutic approaches that specifically address the immune system in patients with FMS.
Further Information
Univ.-Prof. Claudia Sommer, MD is the spokesperson for the Clinical Research Unit KFO5001, ResolvePAIN, and leads the projects NP1 and NP5. Fibromyalgia syndrome is being studied in the second funding period of KFO (NP5). Doctoral candidate Sabine Seefried is researching fibromyalgia syndrome in the research group of Prof. Claudia Sommer.
February 2025
Antibody selection and automated quantification of TRPV1 immunofluorescence on human skin.
Jin Y, Brennecke J, Sodmann A, Blum R, Sommer C.
Scientific Reports 2024 Nov 18; 14(1):28496
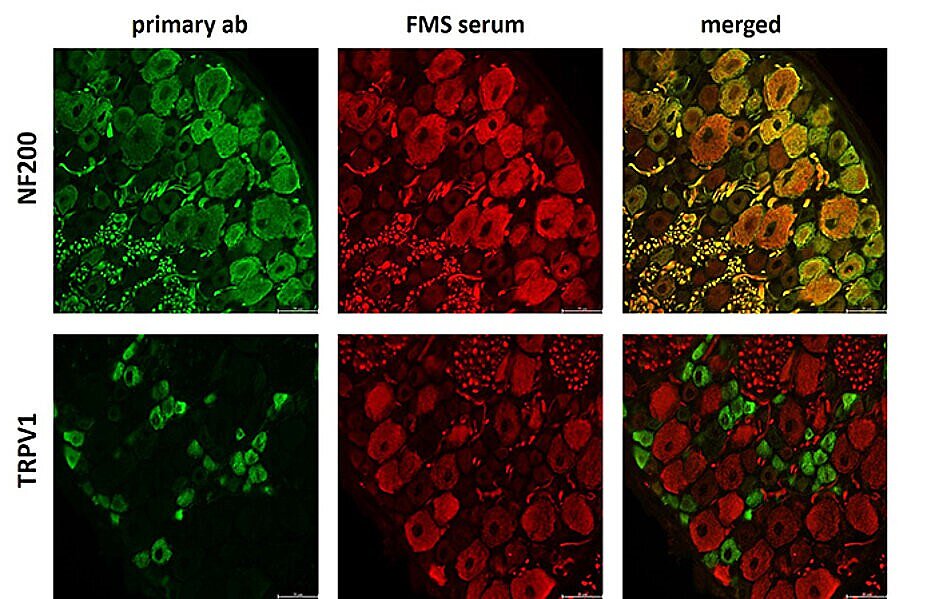
Background
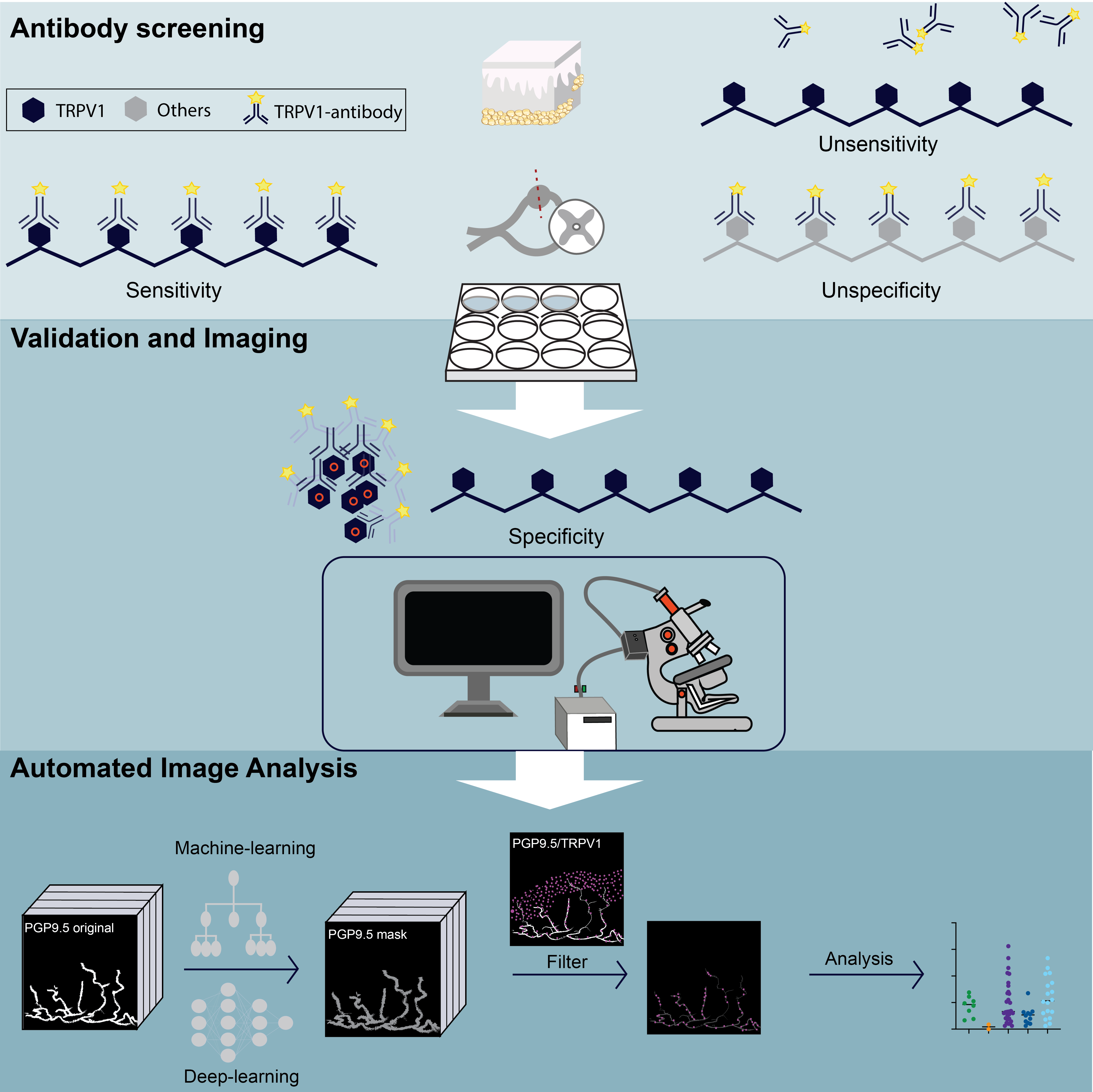
Pain occurs when certain structures in the body perceive and transmit stimuli. The protein TRPV1, which is found in nerve endings, plays a key role in this process. It triggers a burning sensation when exposed to heat and reacts to inflammation or certain chemical stimuli. The receptor is therefore of great importance as a central factor in pain processing and for understanding various pain conditions. Nevertheless, the reliable detection of TRPV1 in human skin has been difficult until now. This was due to unreliable antibodies and manual measurement methods, which led to fluctuations in the results. The aim of our study was to develop a standardised and objective method to reliably detect and quantify TRPV1 in skin.
Experimental Design and Results
We tested six antibodies from different manufacturers to identify those that most reliably detect TRPV1 in skin samples. Using state-of-the-art high-resolution imaging techniques, we visualised the finest structures. We also used advanced machine learning and deep learning to automate the analysis, minimise human error and make the results more reproducible.
It emerged that two specific antibodies recognise TRPV1 particularly reliably. They can be used to precisely measure the quantity and distribution in the skin.
Significance and Perspective
This new method enables the precise detection of TRPV1 in human skin and thus improves the investigation of pain mechanisms in chronic diseases and inflammations. In the long term, targeted therapies could be developed on this basis that block or regulate TRPV1 and thus create new treatment approaches for pain patients. Future studies will now further investigate the role of TRPV1 in various pain disorders and explore ways of specifically influencing its function.
Further Information
Yuying Jin is a doctoral student in the working group of Prof. Dr Claudia Sommer, under whose supervision the study was conducted as part of the Z-Project. Prof Claudia Sommer is also the spokesperson of the clinical research group KFO ResolvePain 5001.
December 2024
Differential effects of everyday-life social support on chronic pain.
Weiß M, Jachnik A, Lampe EC, Gründahl M, Harnik M, Sommer C, Rittner HL, Hein G.
BMC Neurol. 2024 Aug 28;24(1):301. doi: 10.1186/s12883-024-03792-z. PMID: 39198777; PMCID: PMC11351827.
Rationale
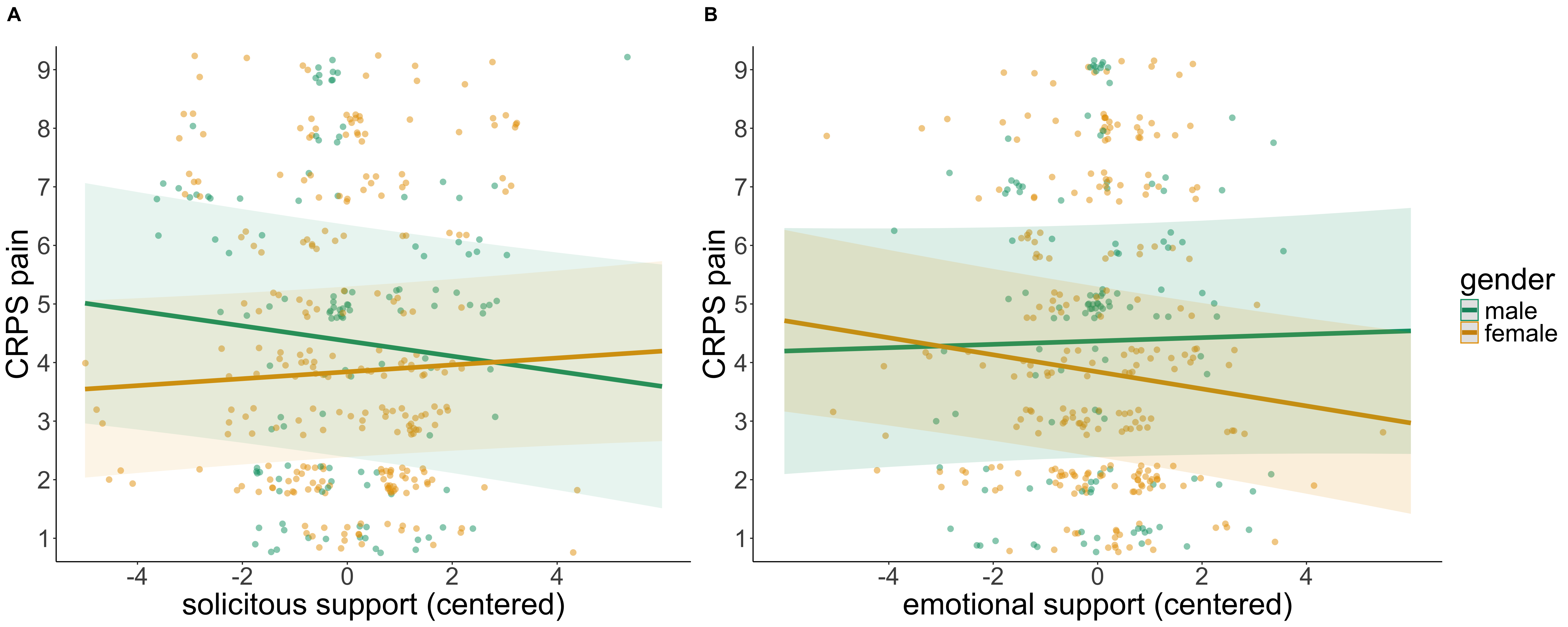
Social support has a complex impact on chronic pain: it can be both alleviating and exacerbating. In daily life, social support manifests in various ways, including emotional support through comfort or attention, as well as practical care, such as taking over tasks. The effects of these different forms of support are debated and have been scarcely investigated, particularly in chronic pain conditions such as Complex Regional Pain Syndrome (CRPS).
Experimental Design
This study used smartphone-based surveys (Ecological Momentary Assessments) to collect data from 20 individuals with CRPS about their daily social interactions. Over five consecutive days, participants received 30 prompts, allowing them to report multiple times a day on the type, frequency, and quality of interactions, as well as the characteristics of the contact persons involved. Simultaneously, they assessed the intensity of their CRPS symptoms, especially pain.
Results
The analysis shows that social support in daily life can significantly reduce CRPS-related pain. Women primarily benefit from emotional support, while men experience pain relief through practical care.
Significance and Perspective
These findings highlight the potential and central role of psychosocial factors in pain therapy and the management of chronic pain. Smartphone-based interventions could potentially be used in the future to target specific forms of support in daily life to contribute to pain alleviation.
Further Information
The CRPS study is part of the Z-Project within the KFO 5001 research group, conducted under the leadership of Univ.-Prof. Dr. med. Heike Rittner. Additionally, Univ.-Prof. Dr. rer. nat. Grit Hein and Univ.-Prof. Dr. med. Claudia Sommer jointly lead Project 5, which investigates social contacts as regulators of pain perception.
October 2024
Greguletz P, Plötz M, Baade-Büttner C, Bien CG, Eisenhut K, Geis C, Handreka R, Klausewitz J, Körtvelyessy P, Kovac S, Kraft A, Lewerenz J, Malter M, Nagel M, von Podewils F, Prüß H, Rada A, Rau J, Rauer S, Rößling R, Seifert-Held T, Siebenbrodt K, Sühs KW, Tauber SC, Thaler F, Wagner J, Wickel J, Leypoldt F, Rittner HL, Sommer C, Villmann C, Doppler K; GENERATE study group.
Different pain phenotypes are associated with anti-Caspr2 autoantibodies.
Journal of Neurology, 2024 May;271(5):2736-2744. doi: 10.1007/s00415-024-12224-4.
Rationale
Patients with Autoantibodies against the protein CASPR2 experience a wide range of symptoms. Some of these patients suffer from pain. This study examined how patients with anti-CASPR2 antibodies differ depending on whether or not they experience pain, as well as the type of pain they experience. These findings could not only provide important insights into the development of the disease but also contribute to more targeted therapies.
Experimental Design and Results
The study included 102 patients with CASPR2 Autoantibodies from a nationwide database in Germany. About one-third of the patients reported experiencing pain, and for more than half of them, the pain was even the primary symptom. The pain could be classified into two groups: one group with burning pain in the feet, and another with muscle ache-like pain throughout the body. No correlation was found between the pain and the antibody titer, antibody subclass, or antibody production in the central nervous system. However, certain risk factors for chronic pain, such as Diabetes mellitus, chronic back pain, or polyneuropathy, were identified as contributing to the development of pain.
Significance and Perspective
Since nerve pain is a common reason for seeking medical attention in neurology, characterizing the type of pain in patients with CASPR2 Autoantibodies can help identify those for whom diagnostic autoantibody testing would be beneficial. Our data suggest that testing for CASPR2 antibodies may be useful in patients with nerve and muscle pain, particularly when accompanied by muscle cramps.
Further information
Dr. Kathrin Doppler (MD) and Prof. Dr. Carmen Villmann jointly lead Project 3, which focuses on the study of neuropathic pain caused by anti-CASPR2 Autoantibodies. Patrik Greguletz is a doctoral candidate working under the Supervision of Dr. Kathrin Doppler (MD) and Prof. Dr. Carmen Villmann in Project 3.

March 2024
Hartmannsberger B, Scriba S, Guidolin C, Becker J, Mehling K, Doppler K, Sommer C, Rittner HL
Transient immune activation without loss of intraepidermal innervation and associated Schwann cells in patients with complex regional pain syndrome
Journal of Neuroinflammation 2023 Jan; 21(1): 23
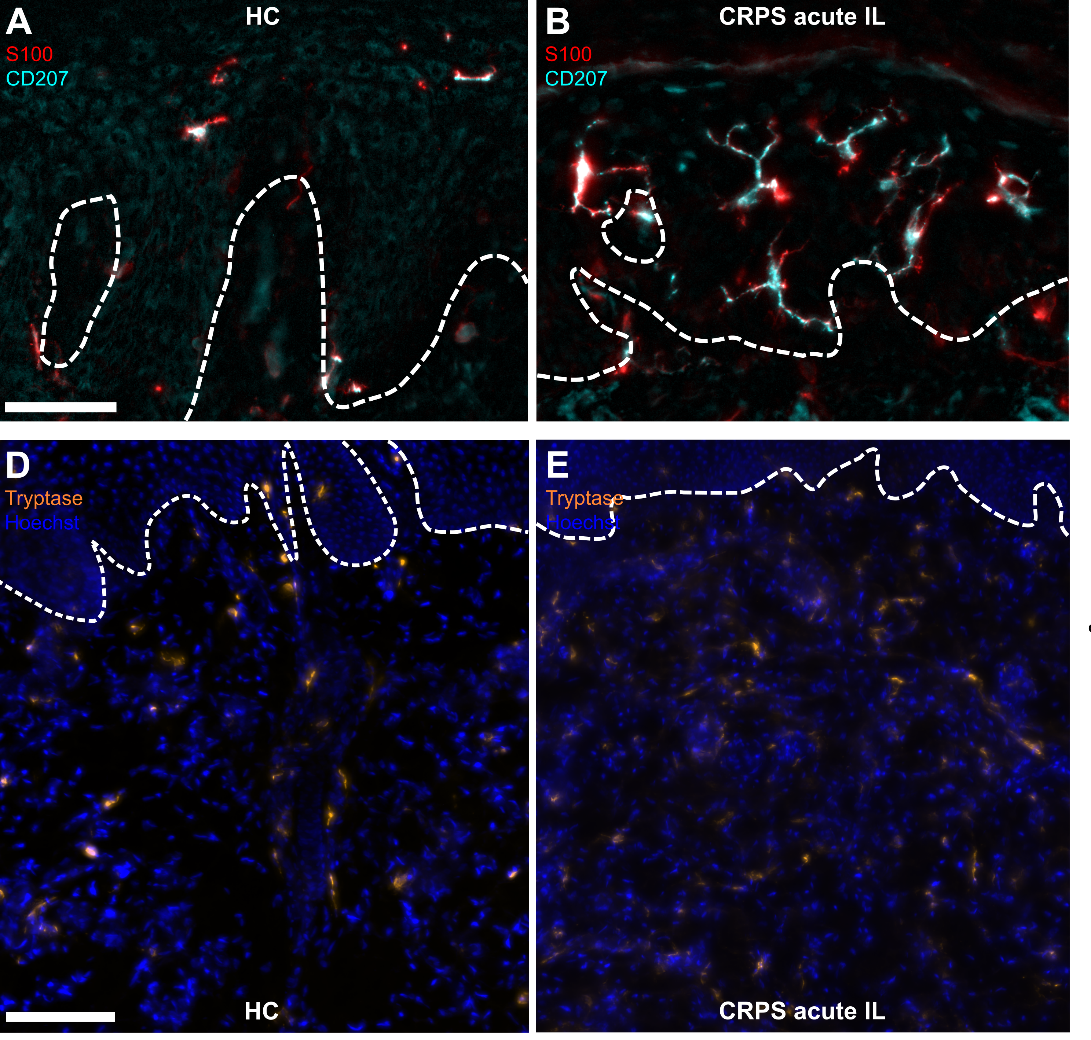
Rationale
Individuals with Complex Regional Pain Syndrome (CRPS) suffer from severe pain and functional impairment of the affected extremity, impacting their quality of life and potentially leading to disability. The precise mechanisms and triggers of this syndrome are not fully understood. Clinically visible symptoms include edema and skin color changes. This study investigated a variety of cell types in the skin that could potentially contribute to the pathophysiology and pain development of CRPS.
Experimental Design and Results
Skin biopsies were taken from patients with CRPS and healthy individuals, and both the nerve fibers and the immune system, as well as the intraepidermal Schwann cells were analyzed and quantified. Notably, Schwann cells were found to accompany the "exposed" nerve endings into the epidermis. Furthermore, in the early stages of CRPS, no nerve degeneration was observed; instead, an immune response involving innate sentinel cells, such as mast and Langerhans cells, was observed. However, this immune reaction pattern was not evident in the chronic stage of the condition. Interestingly, there was an inverse correlation between the number of mast cells and the intensity of pain experienced by patients, whereas no such correlation was observed with the number of Langerhans cells.
Significance and Perspective
The analysis of skin biopsies is a crucial tool for identifying subtypes of CRPS and, consequently, revealing tailored treatment options for this heterogeneous patient group. Our study highlights the importance of early-stage examination for easily identifying existing differences. Further, more detailed analyses will be necessary to precisely understand the mechanisms and, based on that understanding, to develop targeted treatment approaches.
Further information
The CRPS study is part of the Z-project of the CRU 5001. It is being carried out under the supervision of Univ.-Prof. Dr. Heike Rittner, MD.
December 2023
Widder Anna, Reese Lena, Lock Johan Friso, Wiegering Armin, Germer Christoph-Thomas, Kindl Gudrun, Rittner Heike Lydia, Dietz Ulrich, Doerfer Jörg, Schlegel Nicolas, Meir Michael
Postoperative Analgesics Score as a Predictor of Chronic Postoperative Inguinal Pain After Inguinal Hernia Repair: Lessons Learned from a Retrospective Analysis
World Journal of Surgery 2023 Oct; 47(10): 2436-2443
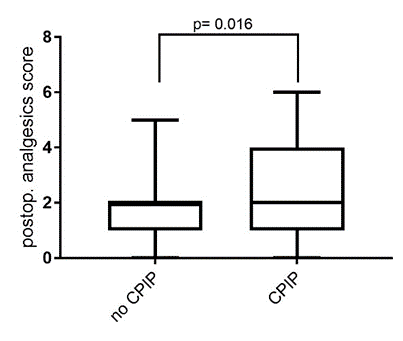
Rationale
In this study, risk factors leading to the development of chronic postoperative pain syndrome (CPIP) were identified in patients who underwent inguinal hernia surgery at the University Hospital Würzburg (UKW) in the last five years. The findings indicate that an increased need for pain medication immediately after surgery, younger age, and female gender are particularly associated with an elevated risk of chronic groin pain.
Study design
Through this retrospective analysis spanning five years, we were able to demonstrate that at the University Hospital Würzburg (UKW), twelve percent of individuals who underwent inguinal hernia surgery developed a chronic pain syndrome. For all participants, a detailed sensory test, morphological imaging, and molecular analysis were conducted to identify objectively measurable parameters through direct comparison.
Outcome and Perspective
Identifying potential changes in patients affected by CPIP compared to healthy individuals provides a deeper understanding of the pathogenesis of this disease. Insights from these findings could potentially lead to new avenues for future therapies. The results of this study are now scheduled for validation in a prospective patient cohort.
Further information
Both, the physician scientist Dr. Anna Widder (MD) and Dr. Michael Meir (MD) work under the supervision of Univ.-Prof. Dr. Nicolas Schlegel (MD) in the Department of General, Visceral, Vascular and Pediatric Surgery (Surgery I) at the University Hospital Würzburg. They are active members of the team studying CPIP in the service project Z.
October 2023
Xiang Zhou, Seungbin Han, Nadine Cebulla, Larissa Haertle, Maximilian J. Steinhardt, Daniel Schirmer, Eva Runau, Leon Flamm, Calvin Terhorst, Laura Jähnel, Cornelia Vogt, Silvia Nerreter, Eva Teufel, Emilia Stanojkovska, Julia Mersi, Umair Munawar, Magnus Schindehütte, Robert Blum, Ann-Kristin Reinhold, Oliver Scherf-Clavel, Heike L. Rittner, Mirko Pham, Leo Rasche, Hermann Einsele, Claudia Sommer and K. Martin Kortüm
Bortezomib induced peripheral neuropathy and single nucleotide polymorphisms in PKNOX1.
Biomarker Research 2023 May 16; 11(1):52
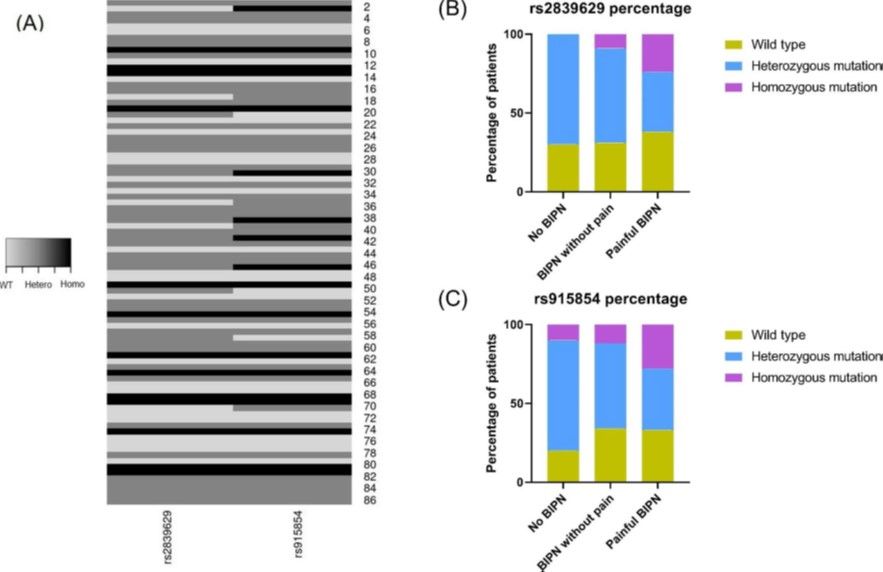
Rationale
Multiple myeloma is the second most common malignant bone marrow disorder in Germany. A crucial component in the treatment of this disorder is bortezomib (BTZ), a well-established chemotherapy drug. Almost all individuals affected by multiple myeloma receive BTZ, however, a significant number of them develop peripheral neuropathy, a dose-limiting side effect of BTZ. The pathomechanisms underlying peripheral neuropathy in these conditions are not yet fully understood. A genome-wide association study (GWA study) suggested that specific nucleotide polymorphisms (SNPs) in PKNOX1 gene correlate with an increased risk of developing bortezomib-induced peripheral neuropathy (BIPN). In the present study, we investigated this possible correlation in myeloma patients treated with BTZ at the University Hospital Würzburg.
Outcome and Perspective
Individuals carrying a homozygous genotype mutation in PKNOX1 (rs2839629) and/or within the intergenic region between PKNOX1 and CBS (rs915854) have a notably higher risk of developing a painful bortezomib-induced peripheral neuropathy (BIPN). Conducting a genetic screening for these specific SNPs before initiating BTZ therapy could offer an improved means of assessing a patient's likelihood of experiencing a painful polyneuropathy, potentially serving as a valuable biomarker for tailoring personalized treatment strategies in the future.
Further information
Information about the authors of this study and the whole project 1 team, led by Prof. Martin Kortum, MD, Prof. Claudia Sommer, MD, and Prof Einsele, can be found on the project's respective side.
May 2023
Cebulla N, Schirmer D, Runau E, Flamm L, Gommersbach S, Stengel H, Zhou X, Einsele H, Reinhold AK, Rogalla von Bieberstein B, Zeller D, Rittner H, Kortüm KM, Sommer C.
Neurofilament light chain levels indicate acute axonal damage under bortezomib treatment
Journal of Neurology. 2023 Feb 18
Rationale
Multiple myeloma (MM) is a plasma cell disorder, responsible for 13% of all hematological malignancies. Bortezomib (BTZ), a well-established chemotherapy drug, is the first-line therapy for this disease. However, despite the efficacy of bortezomib, some patients experience side effects such as Bortezomib-induced peripheral neuropathy (BIPN). Symptoms may regress after the end of the treatment, but BIPN is commonly a dose limiting complication of BTZ treatment, and in some cases, treatment needs to be discontinued. Until now there is no biomarker, which can predict this side effect and its severity. In Project 1 of this Clinical Research Unit, we are investigating the development and regression of peripheral neuropathy and pain under BTZ treatment.
Outcome
In our study, we provided a cross-sectional analysis of our cohort, in which we assessed the levels of neurofilament light chain (NfL). NfL levels were further correlated with several clinical parameters. We found that the axonal damage acutely triggered by BTZ is accompanied by an increase in NfL levels.
Furthermore, these increased levels of NfL correlate with a decreasing amplitude of the nerve action potential on electrophysiological measurements and with the cumulative dose of BTZ: The higher the sum of the BTZ doses administered during therapy, the higher the NfL level and the lower the electrical conductivity of the nerves.
After discontinuing BTZ, NfL levels continually drop over time and approach normal levels again. The next steps of our research are to further investigate the aforementioned alterations in comparison with the clinical course of the disease.
Perspective
We achieved, through the close cooperation of the interdisciplinary and translational team of the KFO 5001, a first step towards understanding BIPN. By analyzing the effect of BTZ on NfL levels, we were able to better characterize and quantify the extent of damage present in BIPN patients. Our aim now is to test whether NfL can be established as a biomarker and be incorporated into daily clinical practice.
Further information
Prof. Dr. Claudia Sommer, MD, is the speaker of the KFO 5001 and one of the principal investigators of Projects 1, 5 and Z.
Nadine Cebulla is a doctoral candidate working under the supervision of Prof. Dr. Claudia Sommer, MD, in Project 1.

March 2023
Appeltshauser L, Junghof H, Messinger J, Linke J, Haarmann A, Ayzenberg I, Baka P, Dorst J, Fisse AL, Grüter T, Hauschildt V, Jörk A, Leypoldt F, Mäurer M, Meinl E, Michels S, Motte J, Pitarokoili K, Stettner M, Villmann C, Weihrauch M, Welte GS, Zerr I, Heinze KG, Sommer C, Doppler K
Anti-pan-neurofascin antibodies induce subclass-related complement activation and nodo-paranodal damage
Brain. Published online Nov 8 2022
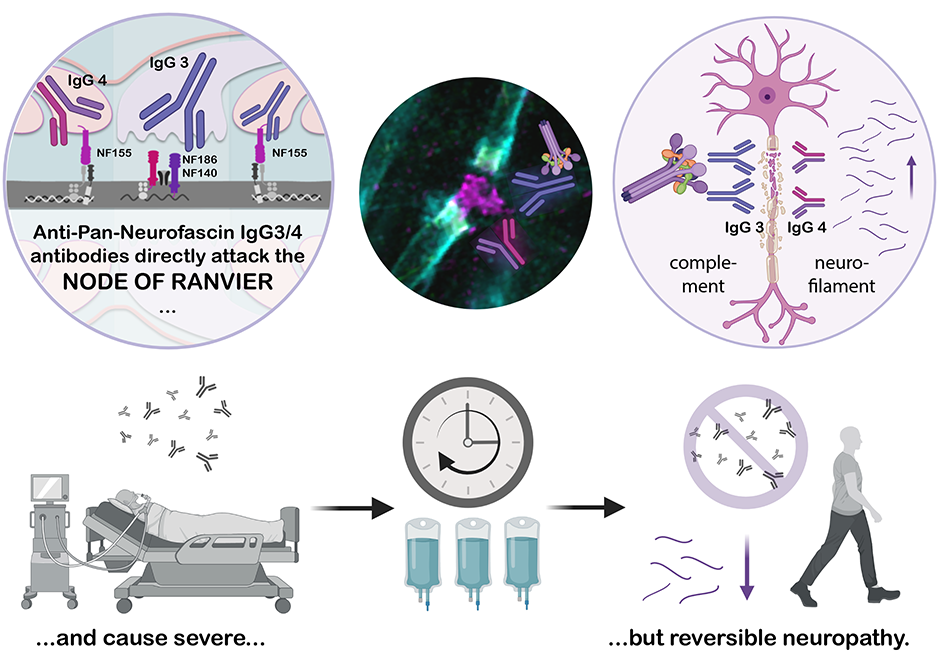
Rationale
In this clinical-experimental study, we described for the first time the effects of pan-neurofascin antibodies on the formation and integrity of the Ranvier Nodes. The Ranvier Nodes, that form gaps within the myelin-sheath, are crucial for the rapid conduction of action potentials. In patients with inflammatory polyneuropathies, neurofascin antibodies destroy the nodal architecture leading to impaired nerve conduction.
Our work includes a detailed description of the clinical consequences of these disorders: Affected patients suffer from an acute and severe polyneuropathy with complete paralysis of the entire musculature, including the respiratory muscles, and often need to be assisted with mechanical ventilation for prolonged periods. However, with immunomodulatory and B-cell-directed therapy, the disease is potentially reversible and usually goes into complete remission after the acute phase.
Perspective
In order to better identify and efficiently treat affected patients, the accurate description of the clinical aspects of this disease is essential to everyday clinical practice. Likewise, understanding the pathomechanism of this disease allows physicians and scientists to use and further develop specific therapies for the disease. Furthermore, a cell culture model established in this study could be easily used in future studies to screen for other antibody-mediated polyneuropathies.
New biomarkers: Antibodies and neurofilament-light chain
In addition to the antibody titer, the authors also identified the neurofilament-light chain as a new biomarker of disease severity, as well as a good marker for assessing the response to long-term therapy. This will help to monitor the disease in a long term, to adjust therapies faster and to identify a possible relapse at an early stage.
Further information
Dr Kathrin Doppler, MD, is the principal investigator of Project 3, in which neuropathic pain caused by anti-Caspr2 autoantibodies is being investigated.
Dr Luise Appeltshauser, MD, works as a Clinician Scientist in the working group of Dr Kathrin Doppler and Univ.-Prof. Claudia Sommer, MD, on autoantibody-mediated neuropathies.
January 2023
Schulte A, Lohner H, Degenbeck J, Segebarth D, Rittner H, Blum R, Aue A
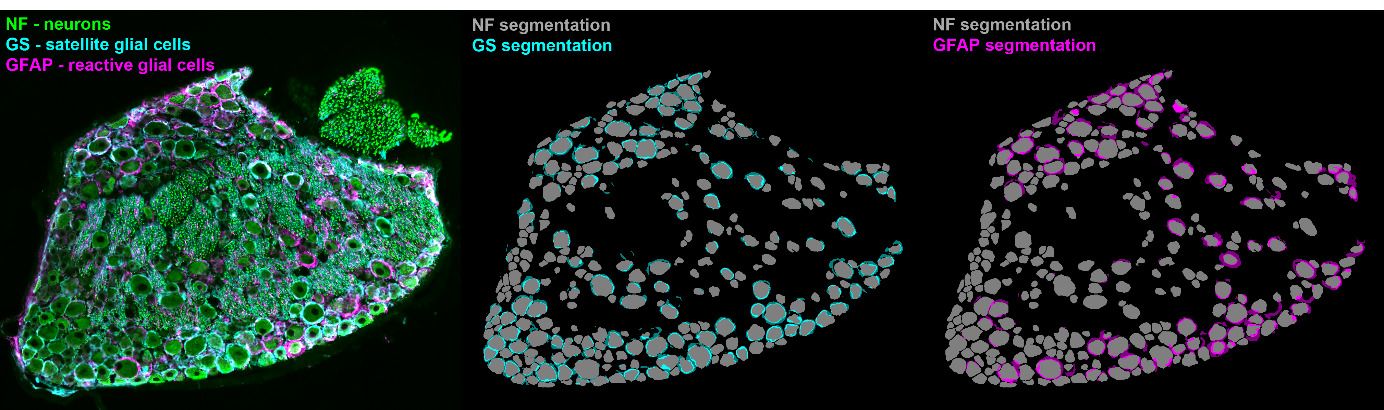
Unbiased analysis of the dorsal root ganglion after peripheral nerve injury: no neuronal loss, no gliosis, but satellite glial cell plasticity
Pain. Published online Aug 15 2022
Rationale
In this paper, we describe the cellular composition of the dorsal root ganglia in an animal model of sciatic nerve injury. For the first time, deep learning (AI-based) image analysis has been applied to a large dataset of microscopy images of dorsal root ganglia. This allowed automatic evaluation of more than 2,500 immunohistochemical images of dorsal root ganglia after nerve injury - something that would have never been possible with previously used methods. As a result, we found that the nerve injury does not lead to a loss of neurons or uncontrolled growth of glial cells, as previously suspected, but only activates the cellular plasticity of glial cells.
Perspective
The data published by our group sets new standards for the objective analysis of microscopic images, here representative in the research area "Molecular Pain Research". This methodology is the basis for describing the multicellular processes of pain resolution with greater precision.
Further information
In project 9 we study the multicellular processes of pain resolution. At present, even cellular processes in human dorsal root ganglia are being studied using deep learning.
November 2022
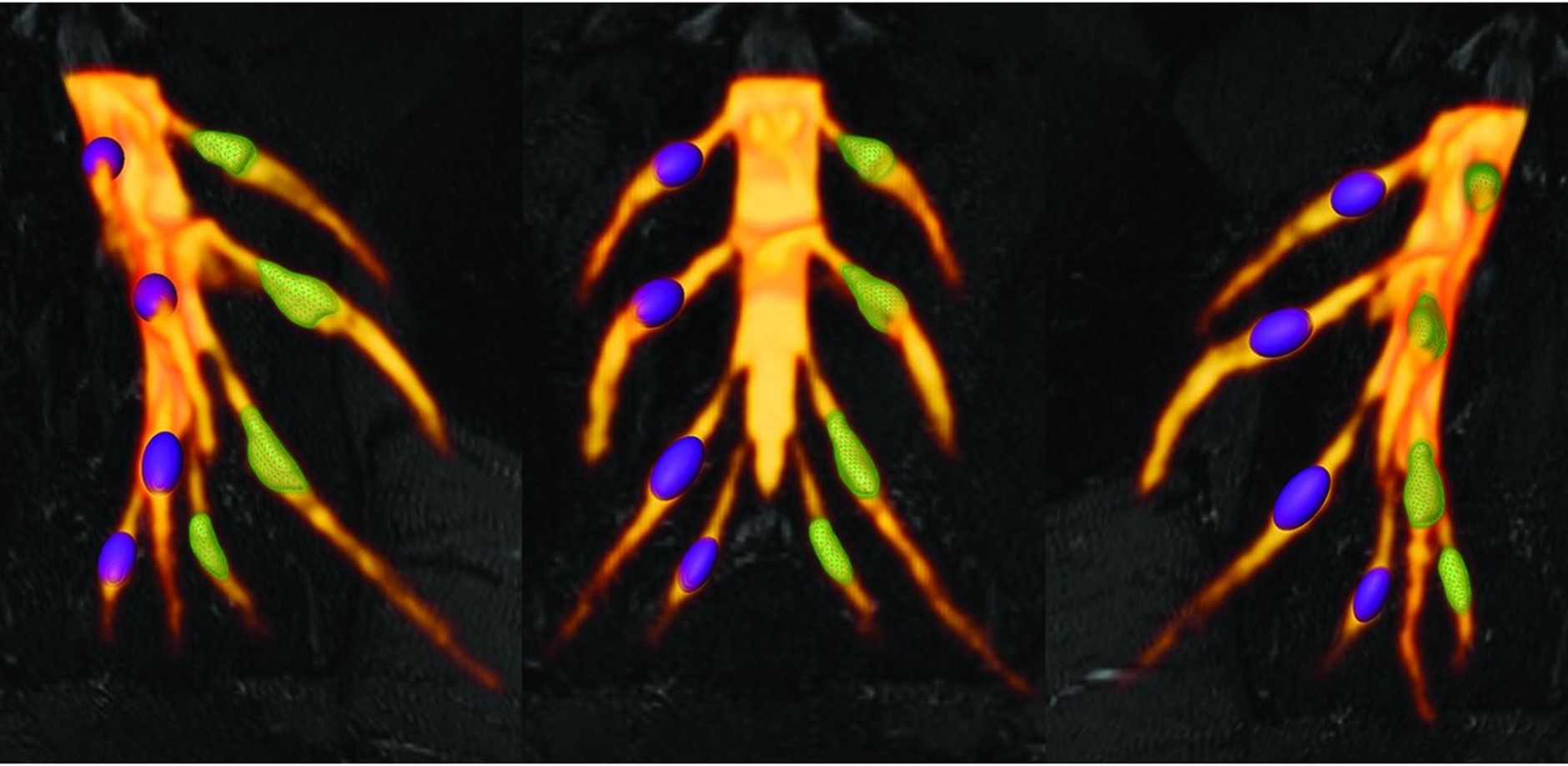
Weiner S, Strinitz M, Herfurth J, Hessenauer F, Nauroth-Kreß C, Kampf T, Homola GA, Üçeyler N, Sommer C, Pham M, Schindehütte M.
Dorsal Root Ganglion Volumetry by MR Gangliography
American Journal of Neuroradiology, Vol 43, No 5 (Mai), 2022: pp 769-775. Epub 2022 Apr 21.
Rationale
Several pain disorders are associated with volume changes of dorsal root ganglia which contain the cell bodies of all sensory peripheral nerve cells. They are significantly involved in pain perception and processing. In this paper, we evaluated a refined method to determine more accurately the volume of the lumbosacral dorsal root ganglia by magnetic resonance imaging (MRI). By ground-truth segmentation, the dorsal root ganglia volume in the individual segments can be determined significantly better and the DRG can be delineated more precise. In parallel, the new method was validated in a cohort of 64 healthy subjects to determine normal values of volume of dorsal root ganglia.
Outlook
The investigation of the dorsal root ganglia using magnetic resonance Gangliography (MRG) provides information on the condition and function of the nervous system in humans. An improved volume determination allows early detection of changes in the dorsal root ganglia. Therefore, on the one hand, the method can play an increasingly important role in the research of painful conditions, and, on the other hand, it can also be used for diagnostics and therapy planning in the care of patients with pain disorders.
Further information
Magnetic resonance imaging of nerves is part of the projekt Z.
Contact
Univ.-Prof. Dr.
Claudia Sommer, MD
PI Project Bortezomib (P1) and Social Environment (P5)
+49 931 201-23763


