Background
Arrhythmogenic cardiomyopathy (ACM) is an inherited cardiomyopathy affecting cell-cell contacts and in particular cardiac desmosomes. Cellular junctions have been shown to be disturbed and non-functional as a result of mutations in desmosomal proteins, but also in response to inflammatory conditions. ACM mutation carriers often present with clinical features of acute myocarditis, suggesting that genetic predisposition and interaction between inflammation and the autoimmune response drive the disease process.
Role of inflammation in ACM remains unclear
Acute and chronic cardiac inflammation has been observed in the clinical presentation of arrhythmogenic cardiomyopathy called “hot phases of disease”. However, the role of inflammation and the immune system in the pathogenesis and the interplay with cellular junction signaling and remodeling needs to be determined.
Aims
The overall goal is to investigate the complex interaction between disturbed cardiac junctions due to genetic defects in
- desmosomal proteins
- cardiac and systemic inflammation and
- autoimmunity
in triggering, maintaining and exacerbating ACM. Additionally, we will analyze, how murine CMV (MCMV) infection and a positive MCMV serostatus affect the development of cardiomyopathy in genetically predisposed heart.
Approach
Assessment of kinetics of cardiac and systemic inflammatory and immune responses via FACS and single-cell RNAseq during cardiomyopathy development in mouse models lacking desmosomal proteins. Determination of the influence of inflammatory cytokines on cell-cell contacts in iPSC derived cardiomyocytes. Investigation of the influence of viral infection as a “second trigger” in a murine CMV infection model.
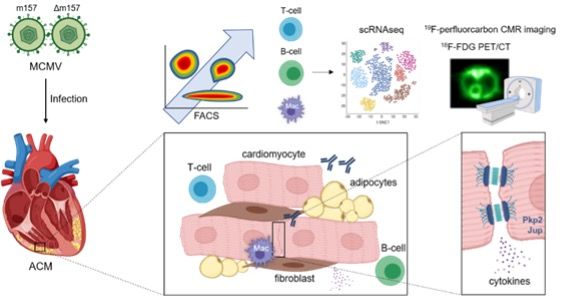
Significance and outlook
We propose that our model-based findings will be translated to humans with the disease towards immunosuppressive and/or anti-inflammatory treatments. We also expect that inflammation and the adaptive immune response may also play a role in other genetically determined cardiomyopathies.
Anschrift
Medizinische Klinik und Poliklinik I, Universitätsklinikum Würzburg, Zentrum für Innere Medizin (ZIM), Oberdürrbacher Straße 6, Haus A3, 97080 Würzburg, Deutschland
Deutsches Zentrum für Herzinsuffizienz Würzburg | Comprehensive Heart Failure Center | Am Schwarzenberg 15 | Haus A15 | 97078 Würzburg


