Role of antigen-specific CD4+ T-cells in myocardial infarction and repair
Lymphocytes are preponderant factors determining the quality of tissue repair in several conditions ranging from skin wound healing to amphibian limb regeneration. However, only recently it became clear that lymphocyte-mediated mechanisms also influence the repair of an infarcted heart. Recent studies from our group and from others revealed that MI triggers the activation of auto-reactive T-lymphocytes, which in turn modulate myocardial inflammation, healing, and fibrosis. Furthermore, pre-clinical studies performed in different labs worldwide proved the principle that cardiac repair can be improved via modulation of lymphocytes’ activity. The big picture is nevertheless far from being clear at the moment, as immune cells interact with the heart in a Janus-faced fashion. Heart-specific immune responses can benefit post-MI healing, but they can also be detrimental in other circumstances like chronic heart failure and aging. Therefore, we need to understand in further details how the immune and cardiovascular systems interact in health and disease.
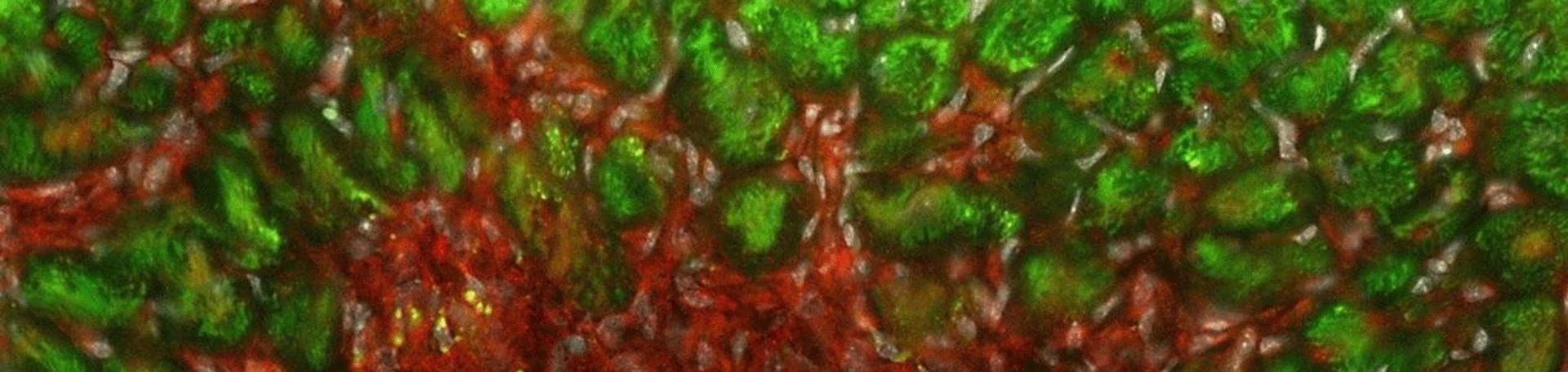
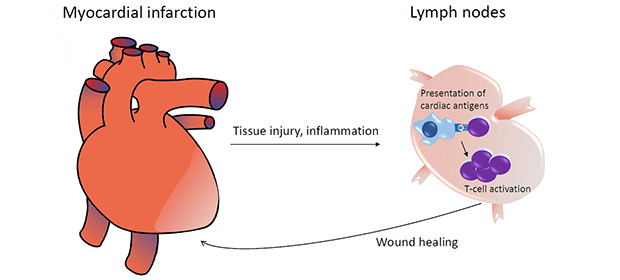
At the Immunocardiology Lab, we approach the infarcted heart from a wound healing-oriented perspective and address how immunological activity influences myocardial repair. Most specifically, we plan to uncover which cardiac antigens are responsible for triggering CD4+T-cell activation in clinical and experimental MI. Furthermore, we plan to dissect the salutary and detrimental aspects of post-MI autoimmunity at both, molecular and systemic levels, so that therapeutic strategies aiming at improving cardiac repair can be eventually designed in the future (Figure 1).
Figure 1: Conceptual framework. Cardiac antigens released by dying cardiomyocytes stimulate T-cell responses which in turn influence post-MI repair. The cardiac antigens, T-cell specificities and phenotypic profile involved in such responses remain largely unknown and are object of study at the Immunocardiology lab. Figure adapted from Ramos, et al., 2016.
Biography
Graduated in Biological Sciences (2005), I developed my PhD thesis at the Federal University of Santa Catarina (Florianopolis, Brazil) focusing on the role of immunological mechanisms in cardiac injury and repair (Ramos, et al., 2012, Doktorväter: Prof. Dr. Jamil Assreuy and Prof. Emer. Dr. Nelson Vaz). Beyond the experimental enterprise, I have also pursued understanding the epistemological aspects of inflammation and immunity (Ramos et al., 2006; 2011; Ramos, 2012). From both, theoretical and experimental perspectives, my major scientific interest has always been to understand how immunological phenomena contribute to body maintenance, similar as to Elie Metchnikoff and Carl Wiegert did more than a century ago. In 2013, I moved to Germany attracted by the “Würzburger School of Cardiology” which is worldwide recognized for its vanguard immunological inclination (Ertl & Frantz, 2005). Together with my colleagues, in the past couple of years I have sought to rediscover the heart under the lenses of immunology, and have contributed to the raising field of Immunocardiolgy (Ramos, et al., 2012; Weirather, et al., 2014; Ramos, et al., 2016; Ramos, et al., 2017; Nunes-Silva, et al., 2017).
Contact Person
Prof. Dr. rer. nat.
Gustavo Ramos, PhD
Leiter des Immunkardiologischen Labors, Heisenberg-Professur für Immunkardiologie
+49 931 201-46477